News

A Brief Discussion on Extracting Rubidium and Cesium from Ore and Its Application
As a minor metal mineral, rubidium and cesium have a relatively small global market consumption, and official production data is lacking. It is estimated that global annual consumption of rubidium and its compounds is in the tens of tons (10-20 t), while global annual consumption of cesium and its compounds is in the thousands of tons (approximately 4,000 t).

A Brief Discussion on the Application of Rare Metals Rubidium and Cesium and Their Compounds
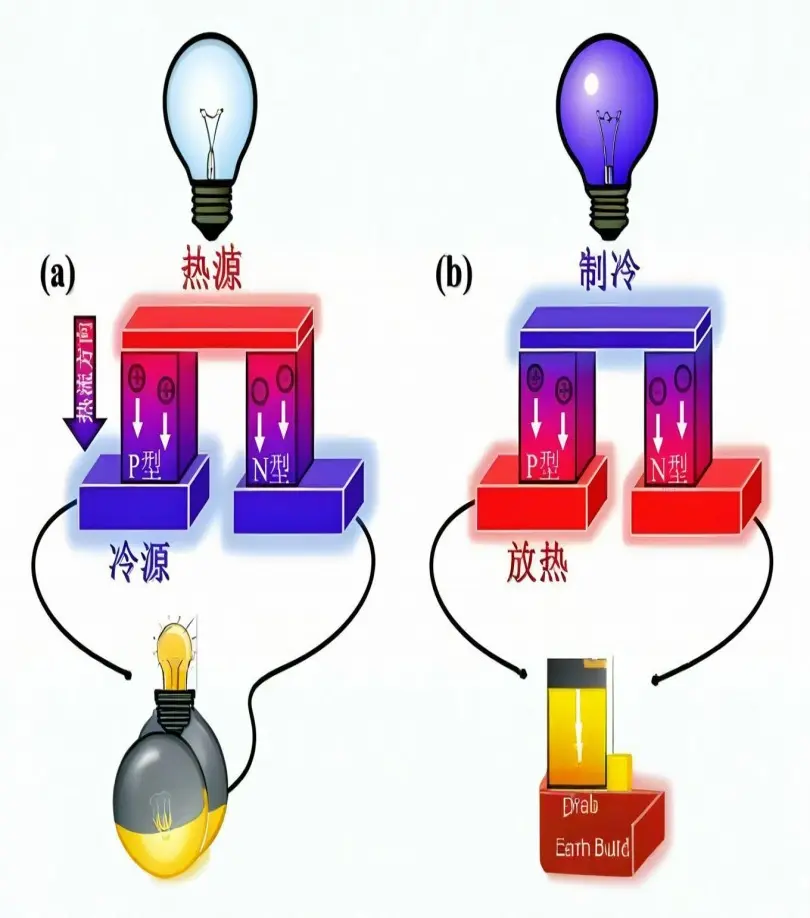
Scientific research has shown that the radiation frequency of rubidium and cesium can remain stable over long periods of time. Atomic clocks produced using rubidium atomic frequency standards utilize transitions between ground state hyperfine energy levels, resulting in advantages such as small size, high precision, low weight, small time error, excellent stability, and low energy consumption.
Rubidium and cesium are chemically active, making them highly ionized. Rubidium is increasingly being used in technologies such as magnetohydrodynamic generators and thermionic conversion power generation. Generators using cesium-containing materials can achieve a total thermal efficiency exceeding 60%. In the aviation industry, rubidium ion propulsion materials used in rockets and spacecraft can achieve a range 150 times that of solid or liquid fuels.

Research and development progress and prospects of high-purity iron preparation
High-purity iron refers to iron with a purity exceeding 99.9%. Iron-based materials developed using high-purity iron as raw material have superior properties compared to traditional iron-based materials, such as lower ductile-brittle transition temperature and recrystallization temperature, improved corrosion resistance, higher maximum magnetic permeability, and excellent electrical and low-temperature thermal conductivity. Therefore, they have broad application prospects in aerospace, electronics, information technology, electric power, military, and other fields.
Application Research of Rare Earth Sulfur Oxides Gadolinium Oxysulfide
Due to their unique layered structure and low phonon energy of approximately 500 cm−1, Rare Earth oxysulfides exhibit extremely high melting points (typically 2000-2200°C) and a narrow bandgap (approximately 4.6-4.8eV). This gives them high chemical and thermal stability, making them frequently used as phosphor matrix materials. These luminescent materials, made from rare earth oxysulfides, have attracted considerable attention for their exceptional light absorption and transmission properties, and have been successfully applied in optoelectronic devices, magnetic materials, catalysts, and other fields.
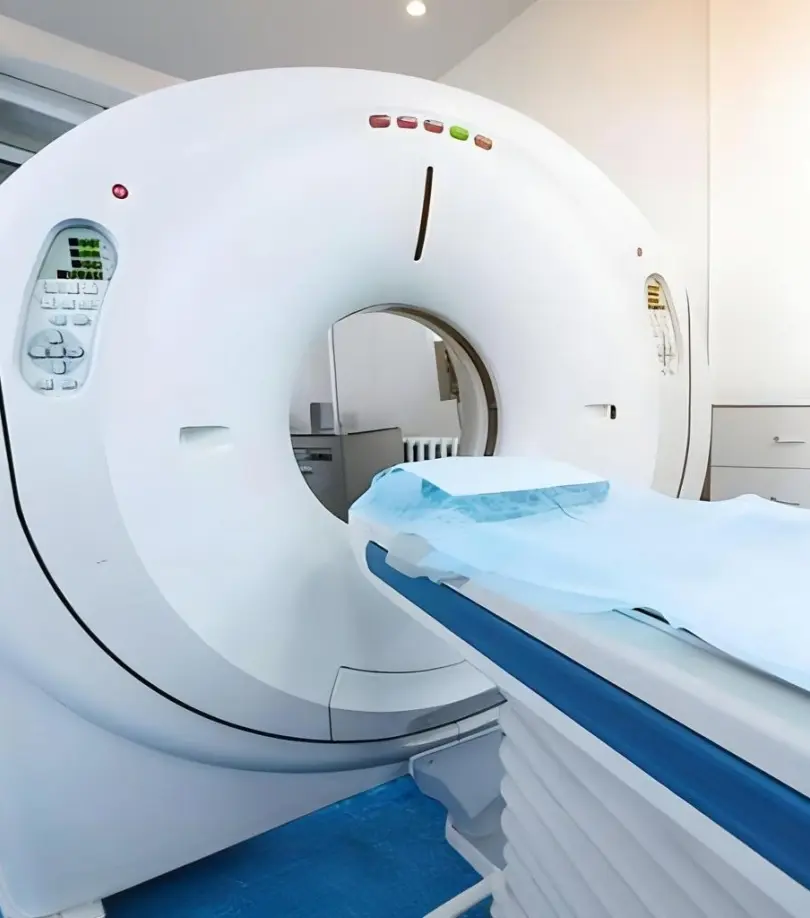
Research on the application of gadolinium oxide in medical contrast agents
Gadolinium contrast agents are currently the most widely used magnetic resonance imaging contrast agents in clinical practice. Gadobutrol, as a second-generation gadolinium contrast agent, is an extracellularly distributed, non-tissue-specific, non-ionic, macrocyclic paramagnetic gadolinium chelator. Due to its high relaxivity, high concentration, and high stability (macrocyclic structure), it has become a leader among contrast agents in combining excellent imaging and high stability.

Application of rare earth oxide gadolinium oxide in uranium-containing pellets
With the increasing demand for economic efficiency in power plants, modern advanced reactors are developing towards large fuel loads, high burnup, and long cycles. This increase in initial fuel load significantly increases the initial residual reactivity of the reactor core within the active zone. In actual operation, separate burnable poison fuel assemblies alone are insufficient to maintain the required long-term reactor reactivity. However, integrated burnable poison fuel assemblies can improve the power distribution within the reactor core, thereby extending refueling cycles and improving fuel economy.

A Brief Discussion on the Preparation of Yttrium Oxide Transparent Ceramics
Yttrium oxide (Y2o3) transparent ceramics have the characteristics of high melting point (2430 ℃), wide optical transparency range (200~8000 nm), excellent corrosion resistance and thermal stability, especially the low infrared emissivity and absorption coefficient exhibited in high temperature environment, which makes it stand out among many transparent ceramic materials and shows broad application prospects in many scenarios such as lasers, transparent windows and semiconductor devices.

Research Progress of Yttria Coatings for Engineering Applications
Yttria coatings, as a key component of rare earth oxide coatings, offer wear resistance, high-temperature stability, and corrosion resistance, playing a vital role in key sectors such as aerospace, electronic communications, and the atomic energy industry. However, given the diverse service environments of engineering materials, traditional Y2O3 thermal barrier coatings are no longer able to meet these complex service requirements. These coatings exhibit poor coating stability, large failure areas, and significantly reduced service life, severely limiting their further adoption in engineering applications.

Research on rare earth flux in recycled aluminum industry
Aluminum and aluminum alloys have a series of advantages such as light weight, high strength, corrosion resistance and recyclability. They are widely used in building materials, decoration, aerospace, automation industry, etc. A large number of aluminum products can be recycled after reaching the end of their service life. Due to the mixture of various grades of aluminum alloys, scrap aluminum contains various impurities and pores. After remelting, the comprehensive mechanical properties of the recycled aluminum alloy are seriously affected and cannot reach the mechanical standards of original aluminum. Therefore, how to regenerate scrap aluminum and make its performance close to or reach that of original aluminum is currently a topic that needs to be studied urgently.

The role of rare earth in structural steel and its research progress
The atomic radius of rare earth is generally between 0.1821 and 0.2042 nm, while the atomic radius of iron is 0.1241 nm. The difference between the two is large, so the solid solubility of rare earth in steel is generally very low. A large number of studies have shown that the addition of trace rare earth elements can significantly improve the plasticity, toughness, wear resistance, heat resistance, corrosion resistance and many other properties of steel.

